Charge of carbon7/2/2023  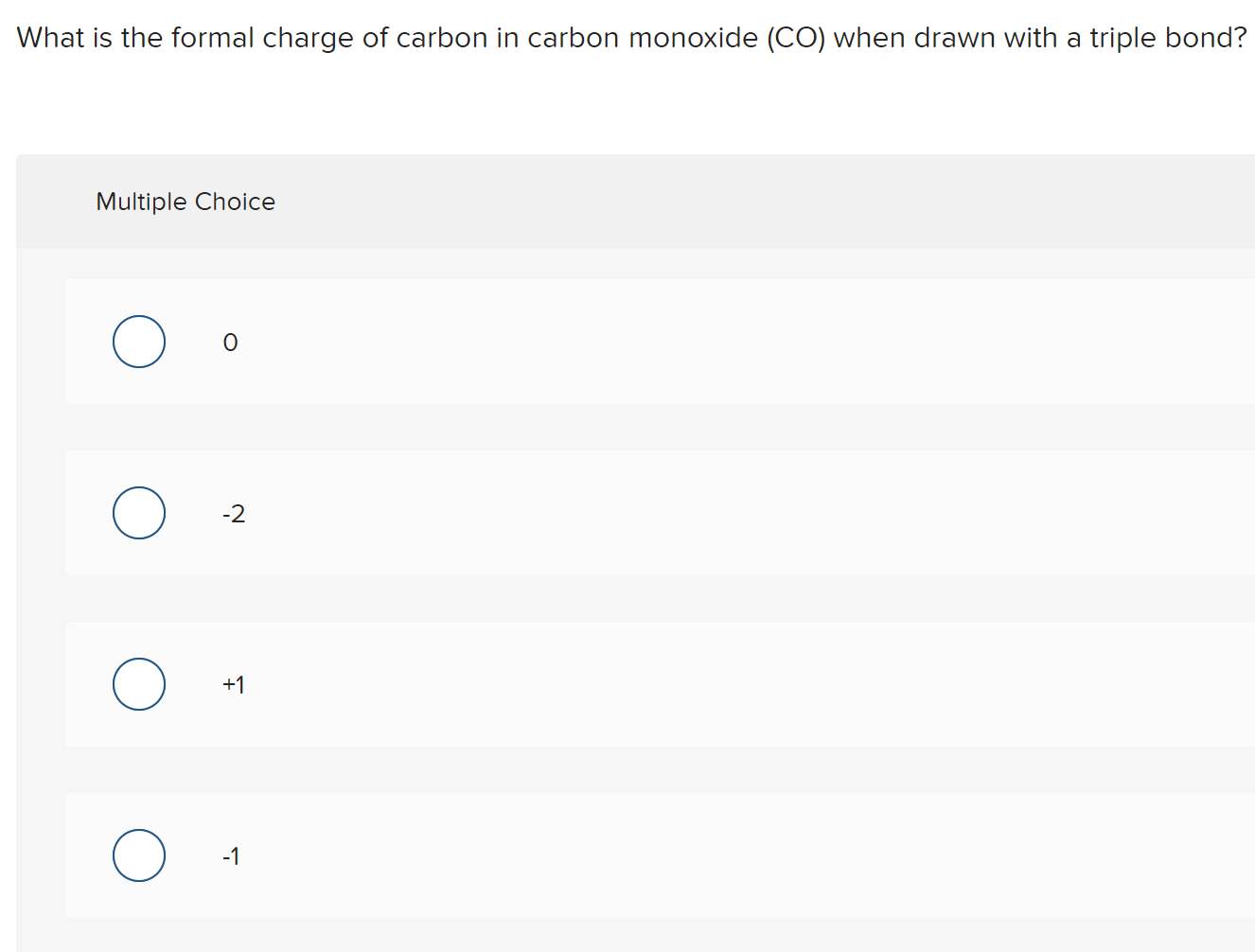 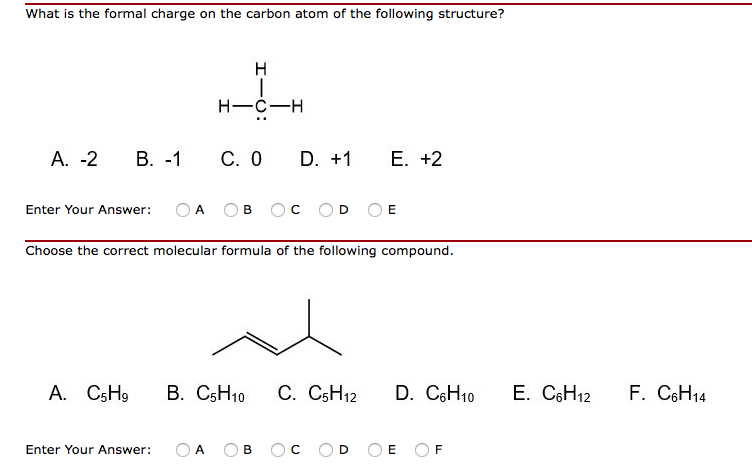
It also describes how these concepts apply to the work that the Department of Energy’s Office of Science conducts as it helps the United States excel in research across the scientific spectrum. DOE Explains offers straightforward explanations of key words and concepts in fundamental science. The recently launched Facility for Rare Isotope Beams has completed its groundbreaking first experimental results.National Isotope Development Center ( Isotope Basics).DOE Isotope Development & Production for Research and Applications.The Journey of Actinium-225: How Scientists Discovered a New Way to Produce a Rare Medical Radioisotope.NSAC: Compelling Research Opportunities Using Isotopes Download Table Nominal and real charges per carbon atom, charge densities and number of ions used in the simulations from publication: Influence of a.NSAC Report: Meeting Isotope Needs and Capturing Opportunities for the Future.Hydrogen is the only element whose isotopes have unique names: deuterium for hydrogen with one neutron and tritium for hydrogen with two neutrons.Some elements can only exist in an unstable form (for example, uranium).All artificial (lab-made) isotopes are unstable and therefore radioactive scientists call them radioisotopes. To provide gold open access, this journal has a publication fee (Article Publishing Charge, APC) which needs to be met by the authors or their research.There are two main types of isotopes: stable and unstable (radioactive).Finally, it conducts research and development on new and improved isotope production and processing techniques. The program also maintains the infrastructure required to produce and supply priority isotope products and related services. The program produces and distributes radioactive and stable isotopes that are in short supply, including byproducts, surplus materials, and related isotope services. The DOE Isotope Program addresses this need. However, isotopes are not always available in sufficient quantities or at reasonable prices. 
Isotopes are needed for research, commerce, medical diagnostics and treatment, and national security. They are important in nuclear medicine, oil and gas exploration, basic research, and national security. Isotopes have unique properties, and these properties make them useful in diagnostics and treatment applications. This decay means the amount of carbon-14 in an object serves as a clock, showing the object’s age in a process called “carbon dating.” Carbon-14 is unstable and undergoes radioactive decay with a half-life of about 5,730 years (meaning that half of the material will be gone after 5,730 years). Carbon-12 is stable, meaning it never undergoes radioactive decay. The addition of even one neutron can dramatically change an isotope’s properties. 
Every element has its own number of isotopes. Carbon occurs naturally in three isotopes: carbon 12, which has 6 neutrons (plus 6 protons equals 12), carbon 13, which has 7 neutrons, and carbon 14, which has 8 neutrons. For example, carbon has six protons and is atomic number 6. The number of protons in a nucleus determines the element’s atomic number on the Periodic Table. Isotopes are members of a family of an element that all have the same number of protons but different numbers of neutrons. Elements have families as well, known as isotopes. The reaction with Br $^-$ is best explained looking at A, though, because the incoming nucleophile ends up bound to carbon, not oxygen.A family of people often consists of related but not identical individuals. The presence of an extra bond in the canonical structure B, along with the completion of the carbon valency makes it more stable than A. And don't you explain reactions with the most relevant contributor, i.e. may have made a mistake while talking about the more relevant contributor, is that when he added Br $^-$ to the compound, he added it to the canonical structure A. Also, won't it prefer to lose out that '+' charge which the bonding's burdened onto it?Īnother thing which made me believe that Prof. Adding up to 8.Ģ) Oxygen being more electronegative than carbon, would pull the electrons towards itself, leading me to think that it would be more comfortable with a lone pair rather than another bond. But I don't understand this.ġ) Isn't the octet complete in A too? It has 2 covalent bonds - 4 electrons (2 from sharing) and 2 lone pairs. Is this true? The reason he gave was that oxygen's octet would be complete in B, hence it contributes more. At first I thought they were wrong, but then the professor agreed with them. I'm watching a video lecture by a professor of my college where he puts this question to the class. Which of the contributing structures of the resonance is more stable? 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
